Phenol extraction
Overview
Phenol (above, carbolic acid) was isolated from coal tar in 1834. It served as a bacteriocide in the late 19th century. Phenol extraction first served to purify (deproteinize) carbohydrates (Westphal, et al. Z Naturforschung B 7, 148, 1952). It was subsequently adapted to "purify" nucleic acids (Kirby, Biochem J 64, 405, 1956). It also separates glycoproteins (Howe, et al, Meth Enzymology 28, 236, 1972) from erythrocyte membrane non-glycoproteins.
Basic principles
Phenol dissolves proteins (for an interesting example see Cohn, E.J. and Conant, J.B. The molecular weight of proteins in phenol. PNAS 12, 433- 438, 1926) and lipids leaving water soluble matter (carbohydrates, nucleic acids, etc.) in the aqueous layer. Particulate and "ambiguous" matter remain insoluble. Phenol extraction of nucleoproteins (e.g virus particles) gives pure products. Crude tissue yields complex mixtures, particularly in the aqueous phase.
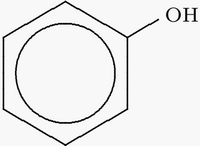
Properties of phenol
Phenol is the simplest aromatic alcohol. Acidity and partial water miscibility are critical for phenol extraction. Phenols are ~ten orders of magnitude stronger acids than aliphatic alcohols and hence vastly better hydrogen bond donors. Amide bonds are stronger bases (hydrogen bond acceptors, same table) than alcohols and ethers. Phenol is partially miscible with water; the aqueous layer contains about 7% phenol and the organic layer about 28% water (Schurmann, E., and Diederichs, R. Thermodynamik des Systems Phenol-Wasser. Berichte der Bunsengesellschaft 68, 434-455, 1964). Above 65C the phases merge. Additives influence phase composition and critical temperature.
Commercial phenol
Phenol can be purchased as a crystalline solid (with 0.1% hypophosphorous acid as antioxidant) or as liquified (88%) phenol. Distillation removes hypophosphorous acid from crystalline phenol or brown coloring (and oxidation products) from liquid phenol. Phenol gradually oxidizes producing a brown color.
Oxidation of phenol
Phenol oxidizes by a free radical process. Apparently copper and phenol contaminants are critical (Dubes, G.R., and Al-Moslih, M.I., Arch Virol 66, 27;1980). Histidine inhibits oxidation (same reference). Judging by Citation Index the Dubes paper (and related papers) has been overlooked. Free radical oxidation cleaves polynucleotide backbones (a basis for synthetic "nucleases"). See "stabilizing liquid phenol."
Preparing phenol
Phenol is often distilled and then saturated with water. Chelators (8-hydroxyquinoline) are added to inhibit oxidation. Phenol is usually neutralized by dialyzing against a buffer. Biochemists have ignored the acidity of phenol (pK ~10) which is itself the major buffer above pH 7 (and will titrate buffers proportional to the buffer/phenol ratio). It is far simpler to neutralize phenol by adding a weak base (such as tris, I prefer N-ethylmorpholine, a liquid). Adjusting the phenol/tris ratio is more convenient (and reproducible) than dialyzing. Raising pH stimulates oxidation so neutralize phenol immediately before use.
Extracting RNA
Tissue is ground with neutralized phenol (usually with a buffer). The aqueous phase is often reextracted with phenol. This helps remove low molecular weight glycosides, etc. RNA is precipitated with ethanol; often following extraction of residual phenol by ether. Smell is a good test for residual phenol; a UV spectrum of an alcohol wash of a pellet is still better. Literature indicates that mRNA is extracted only above pH 7. Since physical properties of RNA do not depend on pH above 5 it is likely that mRNA binding proteins are the culprits.
Extracting DNA
Nuclear DNA has bound histones; higher pH's and salt concentrations (or various additives) help to strip electrostatically bound histones (Kirby, K.S. Biochem J 66, 495-504; 1957)
Removing carbohydrates from phenol extracted nucleic acids
CTAB (cetyltrimethylammonium bromide, Bellamy, A.R., and Ralph, R.K. Meth Enzymology 12B, 156; 1968) is commonly used to precipitate nucleic acids. It leaves neutral carbohydrates in solution. Cetyltrimethylammonium is exchanged out be washing the pellet with an alcoholic salt. The method will obviously not remove anionic polysaccharides.
Phenol additives
Chloroform and aliphatic alcohols are often added to phenol. The latter are antifoams (unnecessary). Chloroform is an antifreeze and increases phenol phase density. 15% chloroform gives enough density to float plant insolubles which stabilize the interphase (convenient). Suggestions that chloroform improves extraction are likely false.
Purifying proteins
Proteins are easily precipitated from phenol with 10 volumes of methanol. I use Ficoll as a carrier. Ficoll stabilizes the pellet and makes it visible. Four volumes of methanol efficiently precipitates all but the smallest proteins. Acetone precipitates phospholipids. Methanol won't precipitate alcohol soluble proteins (usually of little concern). Methanol precipitates proteins poorly from acidic solutions. An organic base or a buffer (e.g. ammonium acetate) solves this problem. Adding sucrose during protein extraction conveniently "inverts" phases.
How efficient is phenol extraction of proteins?
The distribution coefficient of proteins into the phenol phase is greater than 100 (Pusztai, A. Biochem J 99, 93; 1966, in my experience greater than 1000). Hydrogen bonding to the peptide backbone is important; small peptides likely have lower distribution coefficients. Phenol is less a denaturant than a solvent. Salt (or higher pH) helps to extract histones and heat, higher pH or denaturants help to strip TMV capsid from the virion.
Are other phenols better extractants?
Since hydrogen bonding is important, more acidic phenols (e.g. chlorophenol) are likely better extractants, though phenol itself is so efficient that improvements may not be of practical benefit. Hydrophobicity also influences extraction; it is not clear how these forces balance in practice.
Improving nucleic acid and protein extraction
pH and salt concentration are the two easily controllable variables. Salt at acid pH facilitates protein extraction , but higher pH suppresses extraction of organic acids. LiCl is a convenient salt because it's soluble in acetone and alcohols.
General comment on the scientific status of phenol extraction
Phenol has been through two heydays, firstly the turn of the century as a disinfectant and secondly in the '50s as a nucleic acid extractant. One most commonly uses recipes and works with complex systems. Phenol extraction of model compounds has been ignored. Phenol extraction is unfortunately more art than science.